 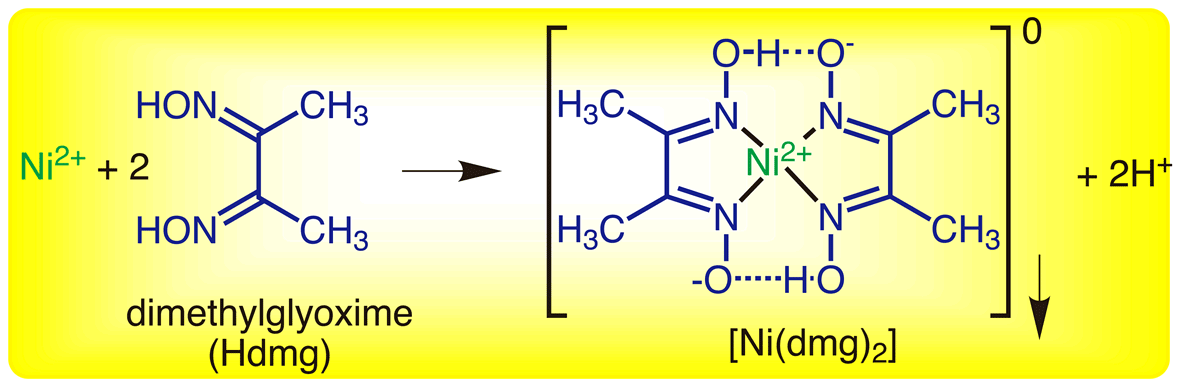
Its coordination complexes are of theoretical interest as models for enzymes and as catalysts. Cyanide salts, their solutions, and hydrogen cyanide gas produced by the reaction of. DmgH 2 is used in the analysis of palladium or nickel. If the ligand is high spin then the arrangement will be different. Pour bottle Four (12.5 mL of 1 dimethylglyoxime) into the beaker. Always consider the spectrochemical series while filling electrons in the orbitals of metal. Note: Remember if there is presence of unpaired electrons then the complex is paramagnetic and if no unpaired electrons are present then, the complex is diamagnetic. So, the electronic configuration will be $\left3\] complex will be diamagnetic in nature. Nickel exists in 2 oxidation state in the complex. 
So, two dmg molecules will donate eight electrons to form the complex. So, it has two donor nitrogen atoms which means one dmg donates four electrons to the nickel atom. NiCl2 in the presence of dimethyl glyoxime (DMG) gives a complex which precipitates in the presence of NH4OH, giving a bright red colour. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
